Today, diagnosing a disease alone is often not enough. Pathologists are increasingly expected to offer insights into prognosis and to predict the most effective treatments. Many of these assessments rely on understanding pathology at a molecular level. Future practice will depend heavily on “omics” technologies, including rapid, cost-effective nucleic acid sequencing, proteomics via mass spectrometry, and immunohistochemistry. Determining when to apply these tests and interpreting their results to guide patient management will be a crucial responsibility for pathologists.
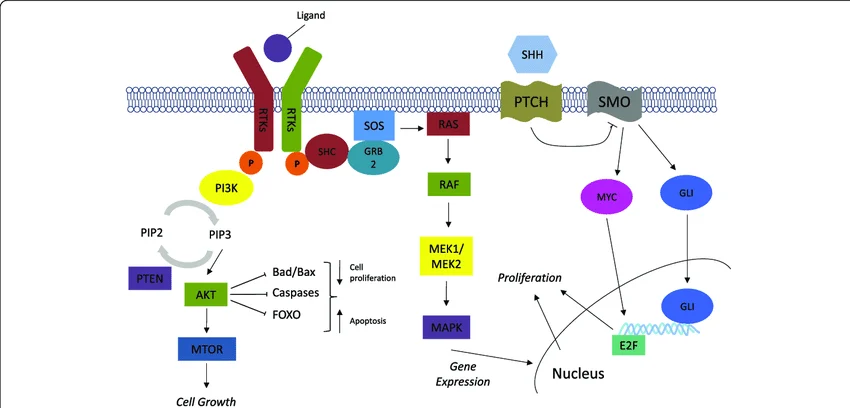
This figure identifies some of the common pathways involved in cancer cell growth and proliferation. Proteins such as Receptor Tyrosine Kinases (RTKs) (EGFR, HER2, VEGFR, PDGFR, IGFR, KIT) PI3K, AKT, RAF, MEK, and SHH represent some of the drug targets in precision medicine.
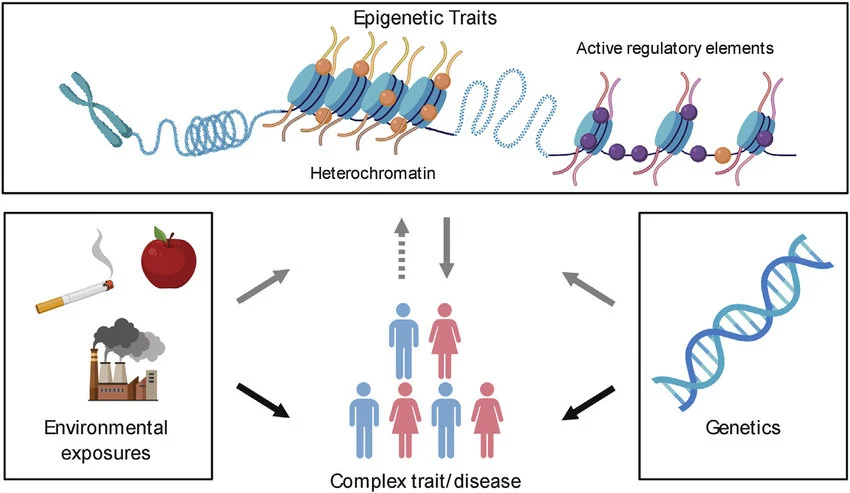
The influence of genetics versus environment varies among diseases. In some, genetic factors are dominant, such as inherited metabolic disorders like phenylketonuria. Rare diseases—approximately 80% of which are genetic—affect about 1 in 17 people in the UK, appearing across many clinical specialties. While an individual may have a disease caused by a single gene mutation, the same inherited disorder can result from mutations in different genes; for example, familial hypercholesterolemia (FH) can arise from mutations in four different cholesterol-related genes. These conditions often show clear inheritance patterns, such as autosomal dominant inheritance, when multiple family members are affected.
Advances in genetics and sequencing have enabled the development of new tests for rare monogenic diseases. Previously, testing was slow, expensive, and focused on single genes selected by genetics specialists. Today, gene panels allow multiple relevant genes to be tested simultaneously, reducing time and cost. In the future, whole genome sequencing (WGS) may allow comprehensive testing across all genes, with results accessible to clinicians in various specialties.
Clinical guidelines, testing criteria, and educational resources support genetic testing, but expert interpretation from clinicians and pathologists remains essential, especially for larger panels or WGS, due to potential variants of uncertain significance. Ethical considerations, including implications for family members, must also be addressed.
Genetics of Common Complex Diseases
Many common diseases such as autoimmune disorders, cancer, and infectious diseases have complex origins, resulting from a combination of environmental risk factors and underlying genetic susceptibility. Advances in medical genetics have improved our understanding of how genetic variation among individuals contributes to disease. Studying these genetic factors not only reveals the biological pathways involved in disease mechanisms, which can serve as potential drug targets, but also shows how rare genetic cases can provide insights into the more common forms of these diseases.
Pharmacogenetics and Treatment
Pathologists play a key role in guiding treatment by using genetic information to understand why patients respond differently to medications. Variations in drug metabolism (pharmacokinetics) or in drug targets (receptors, enzymes, ion channels) can affect efficacy and risk of adverse reactions. Combining genomic data with clinical factors helps predict patient responses, optimize drug selection and dosing, and reduce serious side effects, such as warfarin toxicity. With the growth of whole genome sequencing (WGS), testing for these genetic variants is expected to become increasingly routine in personalized treatment planning.
Cystic Fibrosis and Pharmacogenetics
Cystic fibrosis (CF) is a hereditary disorder that damages the mucous linings of the lungs and digestive system. It is caused by mutations in the CFTR (cystic fibrosis transmembrane conductance regulator) gene. Pharmacogenetics has greatly improved diagnosis and treatment by tailoring therapies to specific genetic mutations.
Drugs such as Ivacaftor and Lumacaftor can partially restore CFTR channel function, while anti-inflammatory medications like NSAIDs and corticosteroids help manage lung inflammation. However, genetic variations in enzymes like CYP2D6 and CYP2C19 can influence drug metabolism, potentially causing adverse reactions.
Pharmacogenetic testing guides personalized therapy: patients with the G551D mutation benefit from Ivacaftor, whereas those with the F508 mutation are treated with combinations like Elexacaftor and Tezacaftor. This approach ensures the most effective and safe drug selection for each patient.
Genetics of Autoimmune Diseases and Vaccines
The complex genetics of autoimmune diseases makes it challenging to determine if vaccines could trigger these conditions. Multiple genes and environmental factors can contribute to a single autoimmune disorder, and genetic markers linked to susceptibility may not be the actual causal genes but are instead closely associated on the DNA strand. These risk factors also vary among different ethnic and racial groups. While it is hypothesized that some adverse vaccine effects could involve viral HERV gene expression, there is currently insufficient experimental evidence to confirm or refute this, making it impossible to attribute vaccine effects to this genetic mechanism at present.
Etrolizumab is a monoclonal antibody targeting the β7 integrin subunit, showing effectiveness in patients with moderate-to-severe ulcerative colitis (UC). Biomarker analysis in the colonic mucosa such as granzyme A and integrin αE, assessed at both gene and protein levels has been linked to clinical response and mucosal healing in UC patients treated with Etrolizumab.
Pharmacogenetic Treatments for Drug Addiction
Disulfiram and methylphenidate pharmacotherapies for cocaine addiction are optimized by considering polymorphisms affecting DβH and DAT1 respectively. Altered subjective effects for amphetamine in DAT1 VNTR variants suggest a ‘protected’ phenotype.
Ethical, Legal, Social, and Organizational Considerations in Genomic Medicine
The implementation of genomic medicine raises several broader challenges, including:
Building genomics expertise across the healthcare workforce.
Addressing patient communication, privacy, and informed consent, especially for testing in children.
Managing uncertain, unexpected, or incidental findings from genomic tests.
Considering the implications of significant results for family members.
Ensuring secure storage, access, and bioinformatics support for genomic data within healthcare systems.
Commencez à écrire ici ...